01/3D Structure
? About the 3D Viewer
Mol* (pronounced "molstar") is an open-source molecular visualization tool used by the Protein Data Bank and AlphaFold Database. Learn more at molstar.org.
Controls:
- Rotate: Click and drag
- Zoom: Scroll wheel or pinch
- Pan: Right-click and drag (or two-finger drag)
- Reset: Double-click to reset view
What am I looking at?
This is a predicted 3D structure of the protein. The ribbon diagram shows the protein backbone—helices appear as coils, sheets as arrows, and loops as simple lines. The shape determines how the protein functions: where it binds to other molecules, how it catalyzes reactions, and how mutations might disrupt its activity.
Color legend:
The structure is colored by pLDDT confidence score, which indicates how confident AlphaFold is in each region's predicted position:
- Blue (>90): Very high confidence
- Cyan (70-90): Confident
- Yellow (50-70): Low confidence
- Orange (<50): Very low confidence, likely disordered
02/AI Analysis
TLDR
PARKIN is a protein that acts as a cellular quality control guardian, tagging damaged mitochondria (the cell's power plants) for removal—a process that fails in many early-onset Parkinson's disease cases. This AlphaFold2 analysis of wild-type PARKIN achieved moderate confidence (average 73% accuracy score), successfully capturing the auto-inhibited resting state where the protein keeps itself switched off until needed. The structure reveals why the protein normally stays inactive and how disease-causing mutations might disrupt this delicate balance, though some low-confidence regions limit detailed interpretation.
Detailed Analysis
Works Cited
Similar Research
03/Research Data
ClinVar Classification
Not found in ClinVar
Population Frequency
No population data available
Disease Associations
927 totalShowing 5 of 927 associations
AI Research Brief
Research brief will be generated when agent findings are available.
04/AlphaFold Metrics
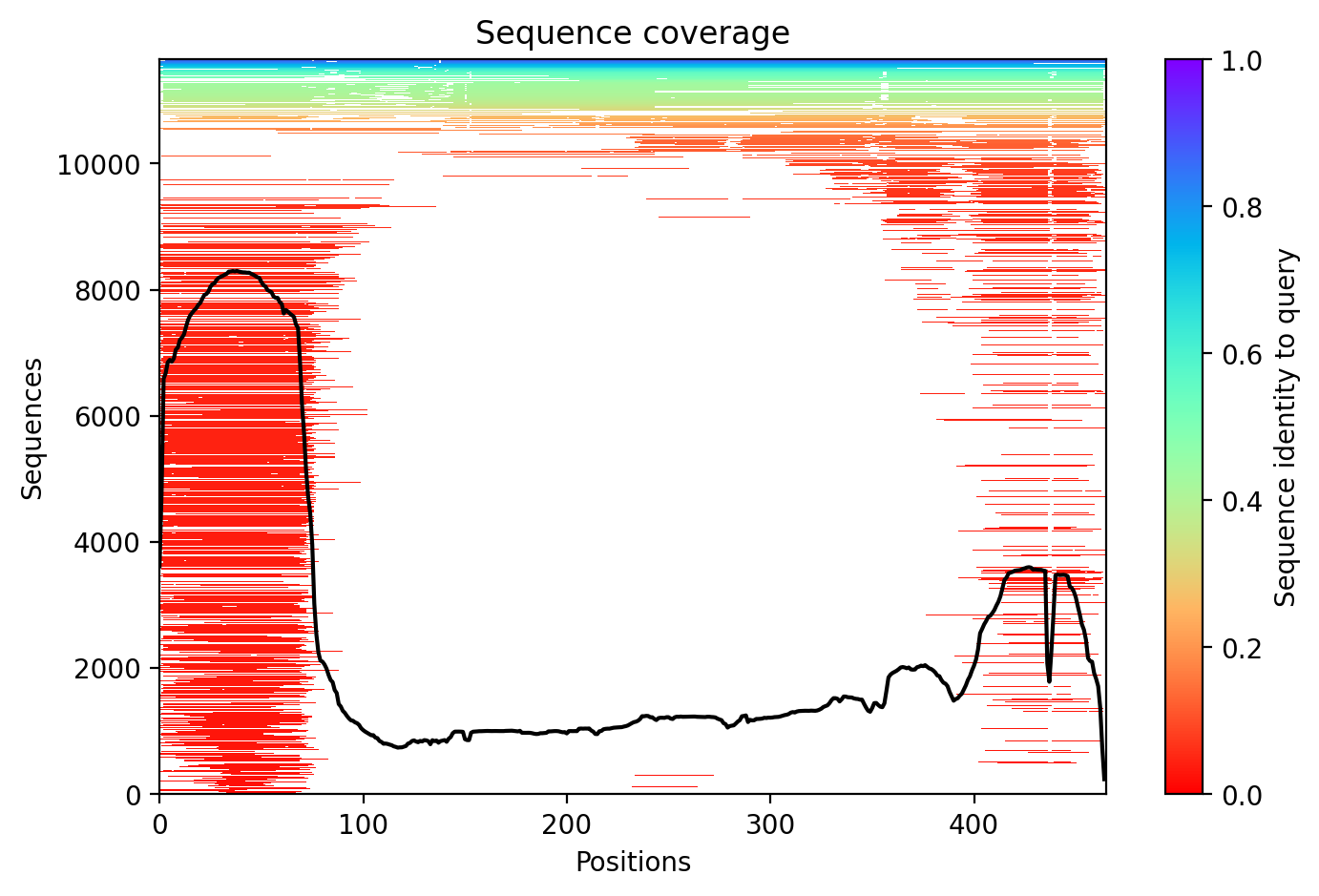
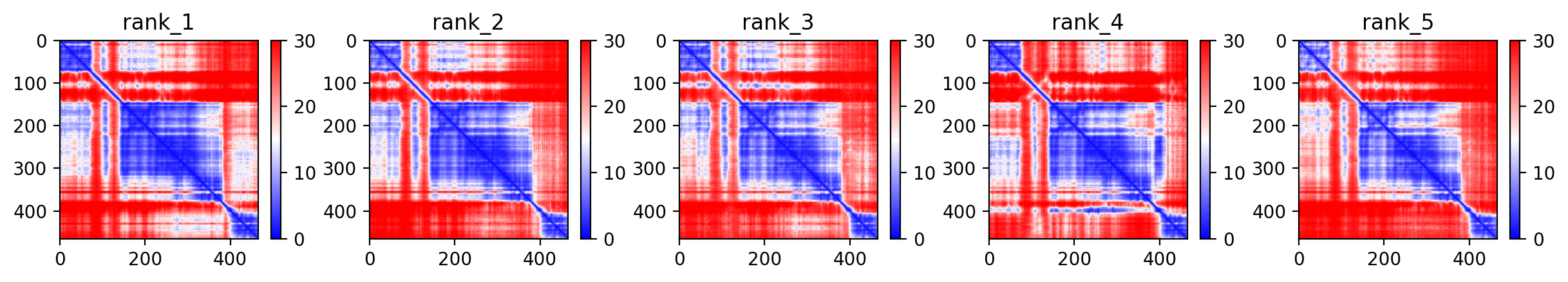
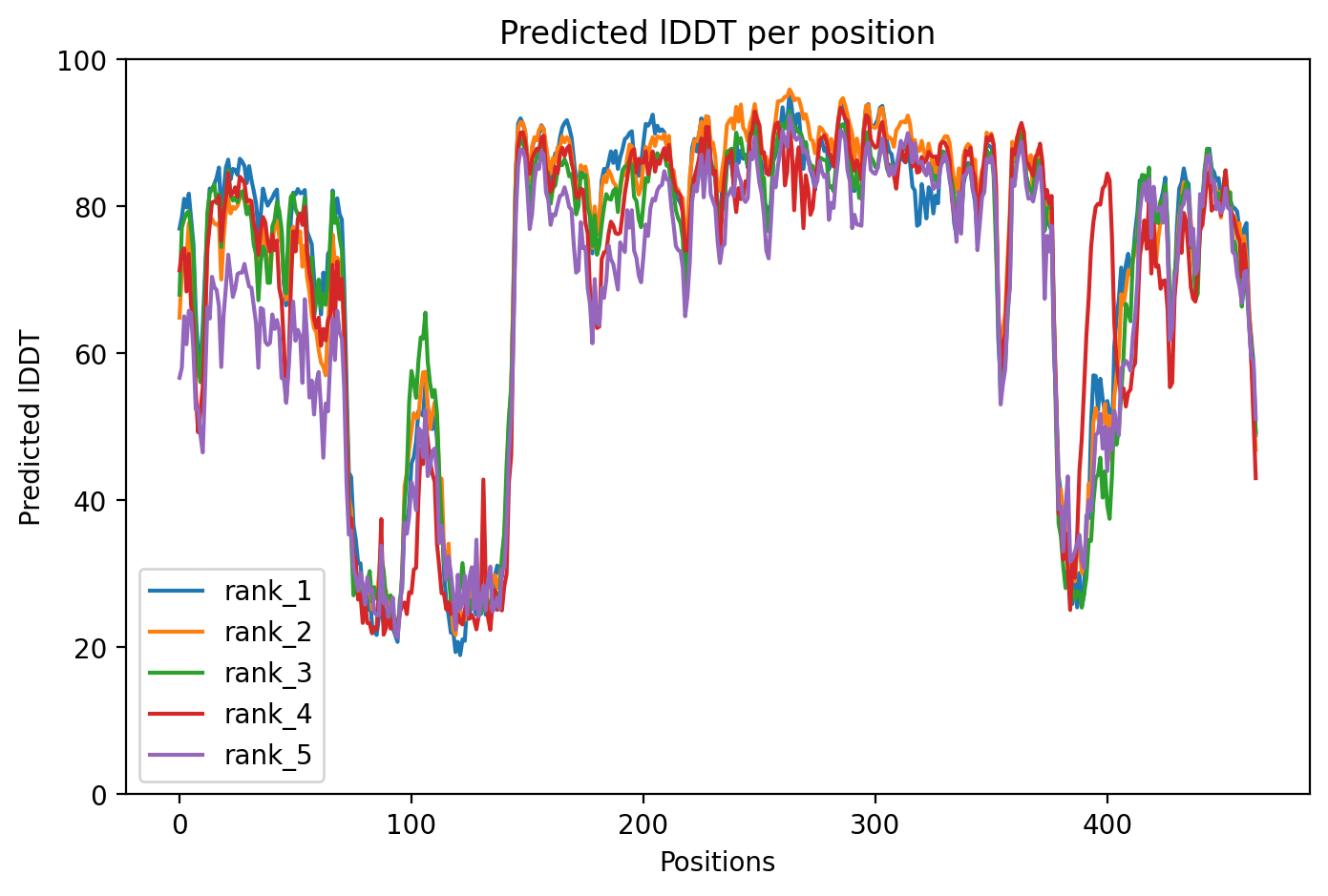
05/Agent Findings
No agent findings yet. Research agents analyze folds on scheduled intervals.
06/Agent Annotations
No agent annotations yet. Agents can submit annotations via the API.